/cloudfront-us-east-2.images.arcpublishing.com/reuters/T3CGA5OL7ZDOTDQFWSJF35D4T4.JPG)
Exclusive: U.S. developing plan to require foreign visitors to be vaccinated, official says
August 19, 2021
Foreign Students Begin U.S. College Deferrals as Delta Spreads
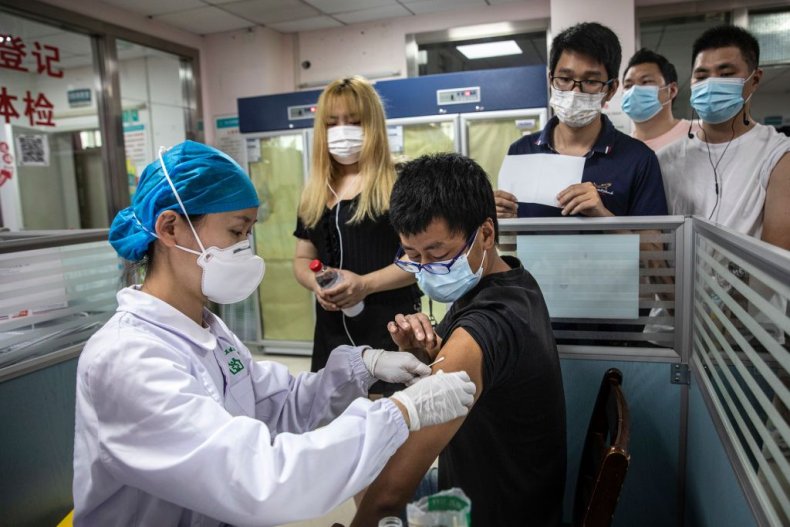
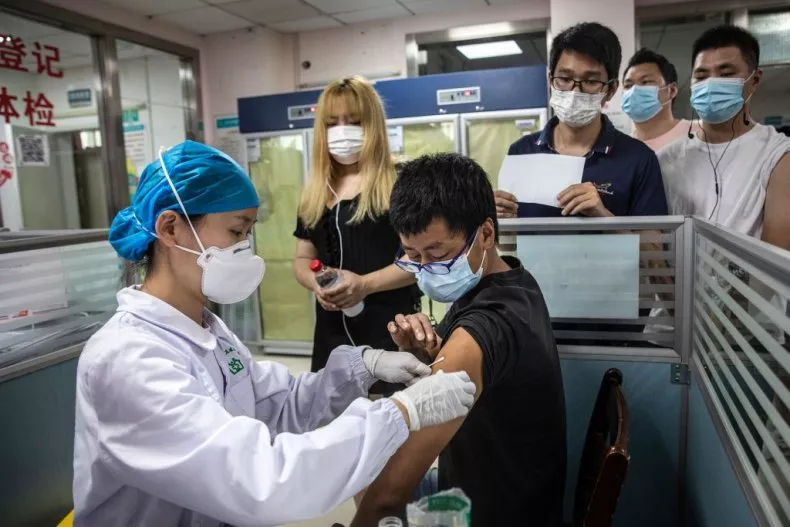
August 25, 2021More than 500 colleges and universities in the United States will require students to be vaccinated against COVID-19 before beginning the fall semester, but a patchwork of rules about which vaccines satisfy the mandates has created uncertainty for international students.
For those who received vaccines in their home countries that are not approved by the U.S. Food and Drug Administration or the World Health Organization, most universities will require another jab. Among the vaccines in this category are those produced in China, Cuba, India, Kazakhstan and Russia.
“We are headed into a very chaotic phase,” Alicia Ely Yamin, senior fellow at Harvard Law School’s Petrie-Flom Center for Health Law Policy, Biotechnology and Bioethics, told Zenger.
Yamin said there is no clear framework in place to evaluate the efficacy of unfamiliar COVID-19 vaccines, and that leaving the decision up to individual institutions is producing “enormous inequities.”NEWSWEEK NEWSLETTER SIGN-UP >
“They need to be able to use governmental and intergovernmental institutions’ approvals as proxies for making their own protocols and policies,” Yamin said.Bloomberg.comSPONSORED BY BLOOMBERG
Subscribe to award-winning journalism and the world’s business news for just $1.99/month.See More

Data maintained by the Chronicle of Higher Education indicate that hundreds of campuses will require students or employees to be vaccinated.
Some colleges, including Boston University, the University of Washington and the California State University system, will accept students who have received any vaccine.NEWSWEEK SUBSCRIPTION OFFERS >
Among the schools accepting only FDA- or WHO-approved vaccines — those produced by Pfizer-BioNTech, Moderna, Johnson & Johnson, AstraZeneca and Sinopharm — are Georgetown University, the University of Chicago and New York University.
“Requiring vaccinations is consistent with our intent to minimize the spread of COVID-19 within our own community, and more generally in New York City, and to enable more in-person on-campus activities,” New York University Provost Katherine Fleming said in an April statement.
Under NYU’s requirements, students who received other vaccines also must quarantine.
“All students who are not fully vaccinated and spend 24 hours or more in a country with a CDC Level 4 Travel notice in the 10 days immediately prior to arriving at NYU will be required to quarantine,” the statement says.
“It makes sense politically,” said Zoe Chen, a New York University student from Beijing, “but individually, I’ll feel like, ‘Why don’t you just admit my vaccine? You are creating more trouble for me.'”
Chen said she has concerns about the safety of repeated COVID-19 inoculations.
“I will have some health concerns because I’m not sure if you can get re-vaccinated in a short period of time,” she said.
Few studies have been done on the safety of mixing vaccine brands.
Early findings from a UK study suggest that it’s safe and effective to mix a “viral vector” vaccine such as the Johnson & Johnson or AstraZeneca shot with an mRNA vaccine like Pfizer‘s. Both vaccines used in the study were approved by the WHO, and research subjects were all older than 50.
The Centers for Disease Control and Prevention says that “people are not recommended to receive more than one complete COVID-19 vaccination series,” but its guidance for those vaccinated outside the U.S. with brands not approved by the FDA or WHO recommends they get a second round.
“You always have to look at any of these vaccines from the perspective of the safety of receiving the vaccine itself and then, will it work?” said Jessica Justman, an associate professor of medicine in epidemiology at the Columbia University Mailman School of Public Health.
Though there are few studies currently available using non-WHO-approved vaccines, the “logic makes sense,” Justman said, since other kinds of vaccines have been shown to be safe together.
“I’d accept getting re-vaccinated,” said Jing Tan, an international student at Stony Brook University on Long Island. Jing received the Sinopharm vaccine, the Chinese-made variant approved by the WHO but not the FDA. “There must be some quality problem if a vaccine is not approved by the FDA.”

China has administered the largest number of COVID-19 shots in the world, just under 1.8 billion doses as of Aug. 16, according to WHO data. Of the six vaccines available there, only three are WHO-approved.
India has distributed the second-highest number, 519 million, with only one of its three brands approved by the WHO.
Justman said that while universities are concerned about whether the vaccines received by returning international students are as effective as those from Pfizer and Moderna, the bigger problem is the limited public acceptance of the shots in general.
“Really, all the authorized vaccines have very, very high rates of efficacy when it comes to preventing severe disease, hospitalization and death,” Justman said.
“The ‘booster’ that the U.S. needs right now is for everyone to get vaccinated.”https://2d6bd6c9c5084b43da9c72662bd546b2.safeframe.googlesyndication.com/safeframe/1-0-38/html/container.html
About 1.1 million international students were enrolled in U.S. institutions of higher education in the 2019–20 academic year, according to the Migration Policy Institute.
This story was provided to Newsweek by Zenger News.
Correction 08/25/21 11:26 a.m. ET A previous version of this story stated that Stony Brook University is in upstate New York. Stony Brook is on Long Island, NY.




56 Comments
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me.
Your article helped me a lot, is there any more related content? Thanks!
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me?
Introducing to you the most prestigious online entertainment address today. Visit now to experience now!
Obrigado|Olá a todos, os conteúdos existentes nesta
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
404085 829670Aw, this became an really good post. In thought I would like to devote writing such as this moreover – taking time and actual effort to make a extremely great article but exactly what do I say I procrastinate alot and by no means uncover a method to get something completed. 264903
**mitolyn**
Mitolyn is a carefully developed, plant-based formula created to help support metabolic efficiency and encourage healthy, lasting weight management.
Can you be more specific about the content of your article? After reading it, I still have some doubts. Hope you can help me. https://accounts.binance.info/en/register-person?ref=JHQQKNKN
I don’t think the title of your article matches the content lol. Just kidding, mainly because I had some doubts after reading the article.
481883 361924I enjoy your writing style actually enjoying this web site . 569716
References:
Muscle building drugs legal
References:
https://platform.joinus4health.eu/forums/users/virgodaniel8/
References:
Casino di sanremo
References:
https://diego-maradona.com.az/user/enginefine69/
References:
Top 5 muscle building supplement
References:
https://www.fcla.de/NEWS/index.php/;focus=STRATP_com_cm4all_wdn_Flatpress_38266970&path=&frame=STRATP_com_cm4all_wdn_Flatpress_38266970?x=entry:entry241215-115425%3Bcomments:1
Thank you for your sharing. I am worried that I lack creative ideas. It is your article that makes me full of hope. Thank you. But, I have a question, can you help me? https://accounts.binance.com/register-person?ref=QCGZMHR6
References:
Steroid girl
References:
https://git.saike.fun:9755/loisbrownlee57
References:
Pros and cons of taking steroids
References:
http://119.29.64.167:3000/edentennant53
References:
Best muscle building drugs
References:
http://132.232.92.186:3000/xsdhayden0265/7422817/wiki/Dianabol%C2%AE+Bodybuilding+Supplement+and+Anabolic+Agent
References:
Physical effects of anabolic steroids
References:
http://101.43.95.130:3001/kirkkenney4529
References:
Where can i buy real dianabol
References:
http://61.145.163.246:3000/lgpgeorgetta82
References:
Steroid for bodybuilding side effects
References:
http://git.chilidoginteractive.com:3000/jeniferainslie/git.fbonazzi.it4342/wiki/Testosterone-Replacement-Therapy%3A-Myths-and-Facts
References:
Steroids legal in australia
References:
http://43.143.175.54:3000/royalholcombe6
References:
Do legal steroids work
References:
https://git.econutrix.com/evangelineeche
First off I would like to say fantastic blog! I had a quick question that I’d like to ask if you do not mind.
I was interested to know how you center yourself and clear your thoughts before writing.
I’ve had trouble clearing my thoughts in getting my thoughts
out. I do enjoy writing however it just seems like the first 10 to 15 minutes are wasted just trying to figure out how to begin. Any ideas or tips?
Kudos!
References:
https://squareblogs.net/wealthtoilet7/das-fuhrende-online-casino-treueprogramm
References:
Instant Casino Kontakt
References:
https://forum.issabel.org/u/harpage0
References:
Gewinne bei Instant Casino auszahlen
References:
https://alushta-shirak.ru/user/lotionshears8/
References:
Instant Casino Auszahlungsdauer
References:
https://f1news.site/item/600776
References:
Instant Casino Konto erstellen
References:
https://diego-maradona-ar.org/user/dangerrotate0/
References:
Play online blackjack
References:
https://lovebookmark.win/story.php?title=winspirit-affiliates-partners-lot
References:
Roulette game download
References:
https://hedgedoc.eclair.ec-lyon.fr/s/2m7M9SZ4F
References:
Online blackjack real money
References:
https://undrtone.com/zebradad34
References:
What is the best muscle builder on the market
References:
https://mcnally-arsenault-2.blogbright.net/clenbuterol-gunstig-online-bestellen-ohne-rezept
References:
Strongest fat burner steroid
References:
https://topspots.cloud/item/595544
References:
Why take steroids
References:
http://uniprint.co.kr/bbs/board.php?bo_table=free&wr_id=272637
References:
What steroids do female bodybuilders use
References:
http://dailyplaza.co.kr/bbs/board.php?bo_table=1302&wr_id=231566
how to use anabolic steroids
References:
https://techniknews.top/item/594052
how are anabolic steroids made
References:
https://undrtone.com/thomasgarden4
References:
Echtgeld Casino Testsieger
References:
https://cameradb.review/wiki/Casino_Apps_im_Test_2026_Die_besten_8_fr_Android_iOS
References:
Mobile casino action
References:
https://analnoe.com/user/crookparent8/
References:
Black jack online
References:
https://jackangel.com/members/daisybrush99/activity/136034/
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.
References:
Buying trenbolone online
References:
https://lyreshow4.werite.net/buy-parabolan-online-safely
References:
Blackjack online game
References:
https://graph.org/Level-Up-Casino-Pro-Tips-for-Maximum-Wins-04-20
References:
Fast payout online https://graph.org/Woo-Casino-Review-Get-Your-Exclusive-Welcome-Bonus-and-Free-Spins-04-20 australia
References:
Fairmont charlevoix
References:
https://android-casino-online.online-spielhallen.de/
References:
Rapunzel video
References:
https://casino-bonus-ohne-einzahlung-2023.online-spielhallen.de/
References:
Mönchengladbach
References:
https://casino-lincoln.online-spielhallen.de/
References:
Ludwigshafen am Rhein
References:
https://casino-marienbad.online-spielhallen.de/
References:
Leipzig
References:
https://spa-and-casino.online-spielhallen.de/
References:
Wolfsburg
References:
https://casino-online-best.online-spielhallen.de/
References:
Ameristar casino st louis
References:
https://graph.org/Jetstar-Oneworld-04-27
References:
Bedste danske casinoer for høje gevinster
References:
http://www.snsopush.com/jeanettthrower
References:
Sikker gevinst udbetaling online casino
References:
http://bsq.cc/bbs/home.php?mod=space&uid=260406
References:
Hvilke online spil har høj udbetaling
References:
https://www.findinall.com/profile/fletalavater04
References:
Hvilket dansk casino udbetaler mest
References:
https://k0ki-dev.com/deonbadham523
Thanks for sharing. I read many of your blog posts, cool, your blog is very good.